|

Source
Journal of Clinical Oncology 2005;23(10):2155-61. Abstract
Purpose
- Evaluate efficacy and tolerability of capecitabine in older women with ABC
Patients and methods
- 73 patients (median age: 73 years old)
- 30 patients received oral capecitabine 1,250 mg/m2 BID on days one to 14
every 21 days
– Due to two toxic deaths, capecitabine 1,000 mg/m2 BID was
administered to the remaining 43 patients
Results
- Dose reductions were required in 30 percent of patients in the standard dose
group, but capecitabine was given without a dose reduction to 95% of
patients in the low-dose group.
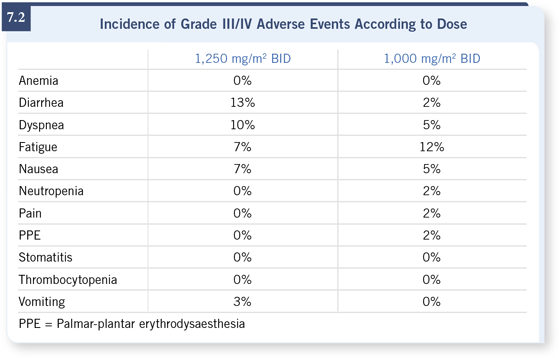
- Capecitabine had a favorable safety profile.
– Grade III/IV toxicities: ≤10 percent fatigue, diarrhea, dyspnea and nausea
- Efficacy
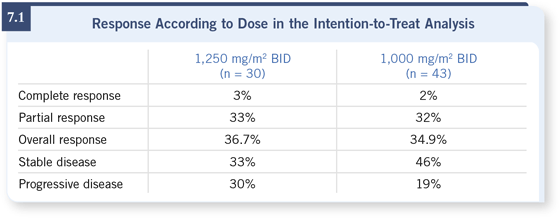
– 1,250 mg/m2 BID: response rate was 36.7 percent; 33 percent had disease
stabilization at ≥24 weeks.
– 1,000 mg/m2: response rate was 34.9 percent; 46 percent had prolonged
disease stabilization.
– Median time to disease progression was four months in both groups.

SELECT PUBLICATIONS
Balducci L et al. Management of breast cancer in the older woman. Cancer Control 2001;8(5):431-41. Abstract
Blum JL et al. Results of a phase II trial of combination therapy with oral capecitabine
and weekly paclitaxel for metastatic breast cancer: Preliminary results in taxane
pretreated women. San Antonio Breast Cancer Symposium 2005;Abstract 6091.
Blum JL et al. Multicenter, phase II study of capecitabine in taxane-pretreated metastatic
breast cancer. Cancer 2001;92(7):1759-68. Abstract
Blum JL et al. Multicenter phase II study of capecitabine in paclitaxel-refractory
metastatic breast cancer. J Clin Oncol 1999;17(2):485-93. Abstract
Di Costanzo F et al. Weekly paclitaxel plus capecitabine in advanced breast cancer
patients: Dose-finding trial of GOIRC and GOL. Ann Oncol 2005;[Epub ahead of print].
Abstract
El-Helw L, Coleman RE. Reduced dose capecitabine is an effective and well-tolerated
treatment in patients with metastatic breast cancer. Breast 2005;14(5):368-74. Abstract
Fumoleau P et al. Multicentre, phase II study evaluating capecitabine monotherapy in
patients with anthracycline- and taxane-pretreated metastatic breast cancer. Eur J Cancer 2004;40(4):536-42. Abstract
Gradishar WJ et al. Capecitabine plus paclitaxel as front-line combination therapy for
metastatic breast cancer: A multicenter phase II study. J Clin Oncol 2004;22(12):2321-7.
Abstract
Hennessy BT et al. Lower dose capecitabine has a more favorable therapeutic index in
metastatic breast cancer: Retrospective analysis of patients treated at MD Anderson
Cancer Center and a review of capecitabine toxicity in the literature. Ann Oncol 2005;16(8):1289-96. Abstract
Holmes CE, Muss HB. Diagnosis and treatment of breast cancer in the elderly. CA Cancer
J Clin 2003;53(4):227-44. Abstract
Kara IO et al. Palmar-plantar erythrodysesthesia due to docetaxel-capecitabine therapy is
treated with vitamin E without dose reduction. Breast 2005;[Epub ahead of print]. Abstract
Lee KS et al. Mature results from a randomized phase III trial of docetaxel/capecitabine
(TX) vs doxorubicin/cyclophosphamide (AC) as primary chemotherapy for patients
(pts) with stage II/III breast cancer (BC). San Antonio Breast Cancer Symposium 2005;Abstract 5052.
Mavroudis D et al. A multicenter phase III trial comparing docetaxel plus epirubicin
versus docetaxel plus capecitabine as first line treatment in patients with locally
advanced and metastatic breast cancer. Preliminary report. San Antonio Breast Cancer
Symposium 2005;Abstract 6089.
O’Shaughnessy J et al. Superior survival with capecitabine plus docetaxel combination
therapy in anthracycline-pretreated patients with advanced breast cancer: Phase III trial
results. J Clin Oncol 2002;20(12):2812-23. Abstract
O’Shaughnessy JA et al. Randomized, open-label, phase II trial of oral capecitabine
(Xeloda) vs a reference arm of intravenous CMF (cyclophosphamide, methotrexate and
5-f luorouracil) as first-line therapy for advanced/metastatic breast cancer. Ann Oncol 2001;12(9):1247-54. Abstract
Reichardt P et al. Multicenter phase II study of oral capecitabine (Xeloda) in patients
with metastatic breast cancer relapsing after treatment with a taxane-containing
therapy. Ann Oncol 2003;14(8):1227-33. Abstract
Schott AF et al. Combination vinorelbine and capecitabine for metastatic breast cancer
using a non-body surface area dosing scheme. Cancer Chemother Pharmacol 2005;[Epub ahead
of print]. Abstract
Talbot DC et al. Randomised, phase II trial comparing oral capecitabine (Xeloda) with
paclitaxel in patients with metastatic/advanced breast cancer pretreated with anthracyclines. Br J Cancer 2002;86(9):1367-72. Abstract
Tong DK et al. Phase II study of an ‘all-oral’ regimen of capecitabine, idarubicin and
cyclophosphamide for metastatic breast cancer — Safety, efficacy and quality of life.
Oncology 2005;68(4-6):520-5. Abstract
Von Minckwitz G et al. Comparison of docetaxel/doxorubicin/cyclophosphamide (TAC)
versus vinorelbine/capecitabine (NX) in patients non-responding to 2 cycles of neoadjuvant
TAC chemotherapy — First results of the phase III GEPARTRIO-Study by the
German Breast Group. San Antonio Breast Cancer Symposium 2005;Abstract 38.
Wagstaff AJ et al. Capecitabine: A review of its pharmacology and therapeutic efficacy in
the management of advanced breast cancer. Drugs 2003;63(2):217-36. Abstract
|

