|

| Tracks 1-14 |
| Track 1 |
Introduction by Dr Love |
| Track 2 |
Integrating aromatase inhibitors into adjuvant therapy of women with ER-positive disease |
| Track 3 |
Importance of quality control in hormone receptor testing |
| Track 4 |
Approach to adjuvant endocrine therapy in postmenopausal women |
| Track 5 |
Management of hormone receptor-positive disease in premenopausal and perimenopausal women |
| Track 6 |
Aromatase inhibitors after five years of tamoxifen in postmenopausal women |
| Track 7 |
Perspective on emerging data on cardiac events in trials of adjuvant aromatase inhibitors |
|
| Track 8 |
Selection of aromatase inhibitors at different timepoints in the adjuvant setting |
| Track 9 |
Approach to and sequencing of therapy of ER-positive metastatic disease |
| Track 10 |
Clinical experience with fulvestrant |
| Track 11 |
Approach to therapy in patients with HER2-positive metastatic disease |
| Track 12 |
Continuation of trastuzumab beyond disease progression |
| Track 13 |
Future directions in adjuvant trastuzumab clinical trials |
| Track 14 |
Implications of accurate HER2 testing |
|
|
Select Excerpts from the Interview
 Track 3 Track 3
 DR LOVE: Would you discuss the issue of quality control in hormone receptor testing? DR LOVE: Would you discuss the issue of quality control in hormone receptor testing? |
 DR VOGEL: I urge all oncologists to take a very close look at their patients with ER- and PR-negative disease because, unfortunately, pathology in the United States has really not kept pace in terms of quality control. DR VOGEL: I urge all oncologists to take a very close look at their patients with ER- and PR-negative disease because, unfortunately, pathology in the United States has really not kept pace in terms of quality control.
We started discovering these problems with one patient in our office who clinically didn’t appear to have ER/PR-negative disease. We sent the slides to Craig Allred at Baylor, and lo and behold, the tumor was strongly ER-positive. So we sent a second and a third. In all, we sent 30 specimens — all read as ER/PR-negative by immunohistochemistry in a broad array of laboratories. Thirty percent turned out to be positive.
 DR LOVE: How much of the problem is technical performance of the assay as opposed to defining the limit for ER positivity? DR LOVE: How much of the problem is technical performance of the assay as opposed to defining the limit for ER positivity?
 DR VOGEL: There are technical problems in some laboratories, and there are cutoff problems in others. The oncologist sees a piece of paper that says “negative” and may not even look at the cutoff values. It is then written in stone in the oncologist’s mind that this patient’s disease is hormonally nonresponsive. We don’t take receptor negativity at face value for any patient. DR VOGEL: There are technical problems in some laboratories, and there are cutoff problems in others. The oncologist sees a piece of paper that says “negative” and may not even look at the cutoff values. It is then written in stone in the oncologist’s mind that this patient’s disease is hormonally nonresponsive. We don’t take receptor negativity at face value for any patient.
 Track 4 Track 4
 DR LOVE: How do you utilize HER2 and PR status in approaching adjuvant hormonal therapy in postmenopausal patients with ER-positive disease? DR LOVE: How do you utilize HER2 and PR status in approaching adjuvant hormonal therapy in postmenopausal patients with ER-positive disease? |
 DR VOGEL: A growing body of evidence indicates that tamoxifen may not be the best hormone therapy in a patient with HER2-positive disease (Ellis 2001). For that reason, I generally choose an aromatase inhibitor for those patients, in both the adjuvant and metastatic disease settings. DR VOGEL: A growing body of evidence indicates that tamoxifen may not be the best hormone therapy in a patient with HER2-positive disease (Ellis 2001). For that reason, I generally choose an aromatase inhibitor for those patients, in both the adjuvant and metastatic disease settings.
 DR LOVE: What about those patients with ER-positive/PR-positive disease? DR LOVE: What about those patients with ER-positive/PR-positive disease?
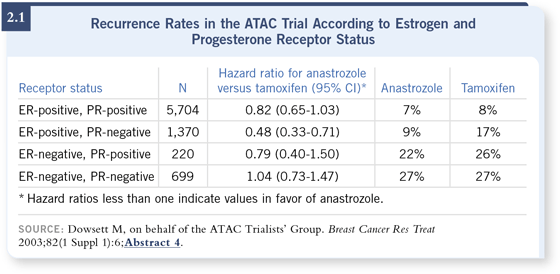
 DR VOGEL: For those patients, we now have another bit of a problem, based on a retrospective subset analysis of the ATAC data from Mitch Dowsett, in which the group of patients with both estrogen and progesterone receptorpositive disease didn’t seem to derive very much efficacy advantage from anastrozole compared to tamoxifen. The greatest benefit was seen in the patients with ER-positive, PR-negative disease (Dowsett 2003, 2005; [2.1]). DR VOGEL: For those patients, we now have another bit of a problem, based on a retrospective subset analysis of the ATAC data from Mitch Dowsett, in which the group of patients with both estrogen and progesterone receptorpositive disease didn’t seem to derive very much efficacy advantage from anastrozole compared to tamoxifen. The greatest benefit was seen in the patients with ER-positive, PR-negative disease (Dowsett 2003, 2005; [2.1]).
However, these same observations have not held up in three other major trials of aromatase inhibitors versus tamoxifen (Coombes 2004b; Jakesz 2005; Goss 2005).
 Track 5 Track 5
 DR LOVE: Would you discuss the adjuvant management of ER-positive disease in the premenopausal patient? DR LOVE: Would you discuss the adjuvant management of ER-positive disease in the premenopausal patient? |
 DR VOGEL: Tamoxifen remains the mainstay of treatment for these patients. Certainly, in Europe there is a very strong feeling that the published data seem to indicate that the addition of ovarian ablation to tamoxifen is superior to either of those modalities alone. In Europe, it’s very hard to convince the vast majority of oncologists that the question of treatment approach in these patients has not already been answered. DR VOGEL: Tamoxifen remains the mainstay of treatment for these patients. Certainly, in Europe there is a very strong feeling that the published data seem to indicate that the addition of ovarian ablation to tamoxifen is superior to either of those modalities alone. In Europe, it’s very hard to convince the vast majority of oncologists that the question of treatment approach in these patients has not already been answered.
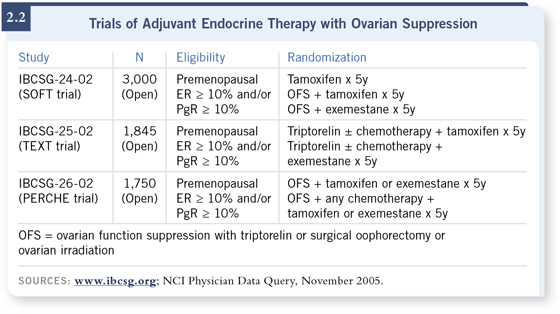
However, the fact that we have the SOFT, TEXT and PERCHE trials examining this very issue indicates that, at least in the minds of most North American oncologists, the question remains unanswered as to the best adjuvant therapy for premenopausal patients (2.2).
The answers are not in and won’t be in for many years. In the meantime, oncologists are stuck deciding what to do. Do you or don’t you believe that the addition of ovarian ablation adds to orally administered hormonal therapy? Certainly, you cannot use an aromatase inhibitor in premenopausal patients and expect it to work unless you render them postmenopausal.
 DR LOVE: What about hormonal therapy for premenopausal women who stop menstruating during or after chemotherapy? DR LOVE: What about hormonal therapy for premenopausal women who stop menstruating during or after chemotherapy?
 DR VOGEL: This must be viewed with considerable caution, especially related to the switching strategy of moving from tamoxifen to an aromatase inhibitor. It is very difficult to follow these women, because the LH and FSH levels are low in tamoxifen-treated patients. You have to rely on the estradiol level, which is fraught with methodological problems. These patients must be followed as closely as possible. DR VOGEL: This must be viewed with considerable caution, especially related to the switching strategy of moving from tamoxifen to an aromatase inhibitor. It is very difficult to follow these women, because the LH and FSH levels are low in tamoxifen-treated patients. You have to rely on the estradiol level, which is fraught with methodological problems. These patients must be followed as closely as possible.
If you want to use the switching strategy in a patient with chemotherapyinduced amenorrhea about whom you’re concerned, you can put her on an LHRH compound and switch to an aromatase inhibitor. I would still monitor her estradiol level for a while.
 Track 6 Track 6
 DR LOVE: Do you believe that five years of tamoxifen followed by an aromatase inhibitor is an acceptable therapy for a postmenopausal patient? DR LOVE: Do you believe that five years of tamoxifen followed by an aromatase inhibitor is an acceptable therapy for a postmenopausal patient? |
 DR VOGEL: I personally might utilize this strategy in a patient with severe osteoporosis, where I am hoping for additional benefit to the bone. In general, however, up front I tend to use aromatase inhibitors as opposed to tamoxifen. DR VOGEL: I personally might utilize this strategy in a patient with severe osteoporosis, where I am hoping for additional benefit to the bone. In general, however, up front I tend to use aromatase inhibitors as opposed to tamoxifen.
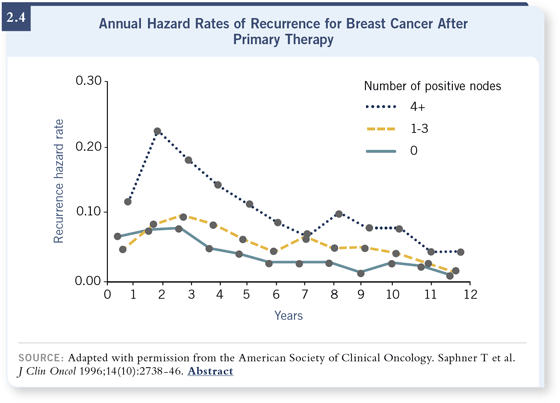
One of the big questions is, do you start with an aromatase inhibitor or do you give the patients two years of tamoxifen? Those who start with aromatase inhibitors are concerned that the first year and a half to two years is one of the major peak recurrence times, and consequently, if you don’t start with an aromatase inhibitor, you will miss that peak recurrence time (Saphner 1996; [2.3, 2.4]). In general, I start with an aromatase inhibitor.
In my postmenopausal patients already on tamoxifen, I generally switch to an aromatase inhibitor after two years. Another unanswered question is, at five years, what do you do with the patients on an aromatase inhibitor? Theoretically and biologically, there is no real reason to discontinue the aromatase inhibitor at five years. We now have the MA17 data, indicating that five years of letrozole after five years of tamoxifen is beneficial (Goss 2003).

 Track 7 Track 7
 DR LOVE: What are your thoughts about the data on cardiac events in patients receiving aromatase inhibitors? DR LOVE: What are your thoughts about the data on cardiac events in patients receiving aromatase inhibitors? |
 DR VOGEL: The first hint came from the BIG FEMTA trial with letrozole (Thürlimann 2005a, b), and they also observed this in the exemestane data (Coombes 2004a). I believe we will see much more in-depth analysis of all of the aromatase inhibitor trials to see just how real this issue is. I don’t know how big an issue the cholesterol problem in the BIG FEMTA trial is because those were not fasting specimens and it was not reproduced in the MA17 trial with the same drug. This is one of those areas where we’re just going to have to wait. DR VOGEL: The first hint came from the BIG FEMTA trial with letrozole (Thürlimann 2005a, b), and they also observed this in the exemestane data (Coombes 2004a). I believe we will see much more in-depth analysis of all of the aromatase inhibitor trials to see just how real this issue is. I don’t know how big an issue the cholesterol problem in the BIG FEMTA trial is because those were not fasting specimens and it was not reproduced in the MA17 trial with the same drug. This is one of those areas where we’re just going to have to wait.
 DR LOVE: Can you put the quantitative risk in perspective? DR LOVE: Can you put the quantitative risk in perspective?
 DR VOGEL: The risk appears to be very low, as we see it at the moment, and the benefits of the drugs appear to be quite high. DR VOGEL: The risk appears to be very low, as we see it at the moment, and the benefits of the drugs appear to be quite high.
We all know about the risk of endometrial carcinoma with tamoxifen, and we have dealt with that. We know about the cardiac risk with adjuvant trastuzumab, and we have to deal with that. There will be many things we need to discuss with patients and document in our charts. For the moment, the cardiac toxicity and impact on lipid profiles with aromatase inhibitors remains to be resolved. More information should become available relatively soon because of the substudies in each of these very large-scale trials.
 Track 9 Track 9
 DR LOVE: What is your approach to patients with ER-positive metastatic disease, both HER2-positive and HER2-negative? DR LOVE: What is your approach to patients with ER-positive metastatic disease, both HER2-positive and HER2-negative? |
 DR VOGEL: To me, any patient with hormone receptor-positive disease is a candidate for hormonal therapy up front, regardless of HER2 status, except those patients with visceral crisis. DR VOGEL: To me, any patient with hormone receptor-positive disease is a candidate for hormonal therapy up front, regardless of HER2 status, except those patients with visceral crisis.
This will be controversial for patients with ER-positive, HER2-positive disease. There are those who feel that a combination of hormonal therapy and trastuzumab should be given up front. I do not fall into that camp, as yet.
To me, every available manipulation we have in metastatic disease is gold. If you use a combination of hormone therapy and trastuzumab, both of which are relatively nontoxic, and you have a two-year response, you really don’t know what would have happened if you had given those agents sequentially. We’re still arguing that point in the chemotherapy arena on the basis of the two Phase III combination chemotherapy trials that claim overall survival benefits without having had sequential arms (O’Shaughnessy 2002; Albain 2004).
 DR LOVE: When you utilize hormonal therapy in metastatic disease, how do you approach the sequencing of the available options? DR LOVE: When you utilize hormonal therapy in metastatic disease, how do you approach the sequencing of the available options?
 DR VOGEL: In postmenopausal patients, for the most part, I generally start with an aromatase inhibitor. There are nine lines of hormonal therapy for postmenopausal women, and there is no tried and true sequence — we don’t have any consensus on a true hormonal cascade. Hormones can be manipulated, in some women, for years. I’ve had patients on hormonal therapy for 10 or 12 years before ever reaching cytotoxic chemotherapy. DR VOGEL: In postmenopausal patients, for the most part, I generally start with an aromatase inhibitor. There are nine lines of hormonal therapy for postmenopausal women, and there is no tried and true sequence — we don’t have any consensus on a true hormonal cascade. Hormones can be manipulated, in some women, for years. I’ve had patients on hormonal therapy for 10 or 12 years before ever reaching cytotoxic chemotherapy.
 Track 10 Track 10
 DR LOVE: What is your clinical experience with fulvestrant? DR LOVE: What is your clinical experience with fulvestrant? |
 DR VOGEL: Fulvestrant is a very good drug that has minimal toxicity. We don’t even encounter much in the way of buttock pain with a five-cc injection. We’re also not seeing the degree of joint discomfort that we see with the aromatase inhibitors. DR VOGEL: Fulvestrant is a very good drug that has minimal toxicity. We don’t even encounter much in the way of buttock pain with a five-cc injection. We’re also not seeing the degree of joint discomfort that we see with the aromatase inhibitors.
In terms of efficacy, fulvestrant seems to be equivalent to anastrozole (Robertson 2003). Based on data published this year in Cancer, there seems to be no difference in overall survival in the randomized trials of anastrozole versus fulvestrant (Howell 2005).
Fulvestrant is a good drug and a viable alternative to aromatase inhibitors in patients who have disease progression on tamoxifen. We do have to contend with the randomized trial of fulvestrant versus tamoxifen, where we expected a strongly beneficial effect for fulvestrant over tamoxifen, which was not forthcoming. There were some subsets where fulvestrant appeared to be better, but the overall results were about the same (Howell 2004).
 Track 11 Track 11
 DR LOVE: Can you summarize your approach to first-line therapy in patients with HER2-positive metastatic disease? DR LOVE: Can you summarize your approach to first-line therapy in patients with HER2-positive metastatic disease? |
 DR VOGEL: First of all, if I don’t have a FISH assay, I won’t treat the patient. Many still feel that an IHC 3+ is absolute, but I disagree. Quality control is improving, but it is still nowhere near where it needs to be. I insist that my patients have a FISH assay before I embark on trastuzumab therapy in the metastatic setting. DR VOGEL: First of all, if I don’t have a FISH assay, I won’t treat the patient. Many still feel that an IHC 3+ is absolute, but I disagree. Quality control is improving, but it is still nowhere near where it needs to be. I insist that my patients have a FISH assay before I embark on trastuzumab therapy in the metastatic setting.
Since I published the paper on single-agent trastuzumab (Vogel 2002), everybody thinks that that is my first-line treatment in every patient. It’s not. Certainly, I believe a course of single-agent trastuzumab is not unreasonable in relatively asymptomatic patients with minimal disease burden. However, for more symptomatic patients, I tend to use a combination of chemotherapy and trastuzumab.
 Track 12 Track 12
 DR LOVE: What do you consider an indication to discontinue trastuzumab in the metastatic setting? DR LOVE: What do you consider an indication to discontinue trastuzumab in the metastatic setting? |
 DR VOGEL: It’s difficult to get patients to stop trastuzumab. I tend to continue trastuzumab almost indefinitely as we switch from chemotherapy to chemotherapy. Is that supportable by data? Absolutely not. Will continuation of trastuzumab beyond progression on first-line therapy ever be proven? My suspicion is not. This may be one of those questions that are never answered. DR VOGEL: It’s difficult to get patients to stop trastuzumab. I tend to continue trastuzumab almost indefinitely as we switch from chemotherapy to chemotherapy. Is that supportable by data? Absolutely not. Will continuation of trastuzumab beyond progression on first-line therapy ever be proven? My suspicion is not. This may be one of those questions that are never answered.
 DR LOVE: How will you approach the new generation of patients who develop metastatic disease after receiving adjuvant trastuzumab? DR LOVE: How will you approach the new generation of patients who develop metastatic disease after receiving adjuvant trastuzumab?
 DR VOGEL: I would use trastuzumab in these patients for the same reasons that we continue it in the metastatic setting. First, we’re still hoping for therapeutic synergism with the next drug that we use. Second, it’s relatively nontoxic and so relatively easy to continue. And the third reason is that preclinical data seemed to indicate that even in rats progressing on trastuzumab, the rate of progression was slowed in those who continued trastuzumab. DR VOGEL: I would use trastuzumab in these patients for the same reasons that we continue it in the metastatic setting. First, we’re still hoping for therapeutic synergism with the next drug that we use. Second, it’s relatively nontoxic and so relatively easy to continue. And the third reason is that preclinical data seemed to indicate that even in rats progressing on trastuzumab, the rate of progression was slowed in those who continued trastuzumab.
Select publicaitons
|

