|

| Tracks 1-12 |
| Track 1 |
Introduction by Dr Love |
| Track 2 |
Background and rationale for
development of the Oncotype
DX™ assay |
| Track 3 |
Oncotype DX assay to predict
prognosis for patients with early
breast cancer |
| Track 4 |
Utilization of the Oncotype DX
assay in clinical practice |
| Track 5 |
Perspective on the economics of
cancer treatment |
| Track 6 |
Recurrence score as a predictor
of response to therapy |
| Track 7 |
Integration of Oncotype DX assay
into clinical practice guidelines |
|
| Track 8 |
Results of NSABP-B-32 sentinel
node trial |
| Track 9 |
Sentinel lymph node biopsy:
Clinical issues in utilization |
| Track 10 |
Clinical use of adjuvant aromatase
inhibitors in postmenopausal
women |
| Track 11 |
Planned NSABP trial evaluating
optimal duration of aromatase
inhibitors |
| Track 12 |
Aromatase inhibitors for
chemoprevention and treatment
of DCIS |
|
|
Select Excerpts from the Interview
 Track 2 Track 2
 DR LOVE: Can you discuss the first major presentation of the Oncotype DX assay NSABP data by Dr Soon Paik at the 2003 San Antonio Breast Cancer Symposium? DR LOVE: Can you discuss the first major presentation of the Oncotype DX assay NSABP data by Dr Soon Paik at the 2003 San Antonio Breast Cancer Symposium? |
 DR MAMOUNAS: The initial study looked at the value of the recurrence score as it was developed based on the data sets from Rush-Presbyterian in Chicago (Cobleigh 2003), St Joseph Medical Center in California (Esteban 2003) and the tamoxifen-treated patients in NSABP-B-20 (Paik 2003). DR MAMOUNAS: The initial study looked at the value of the recurrence score as it was developed based on the data sets from Rush-Presbyterian in Chicago (Cobleigh 2003), St Joseph Medical Center in California (Esteban 2003) and the tamoxifen-treated patients in NSABP-B-20 (Paik 2003).
By putting the data in a multivariate analysis, we found genes that were the most predictive of recurrence; 16 cancer-related genes and five reference genes ended up being the most predictive. So a 21-gene index was developed.
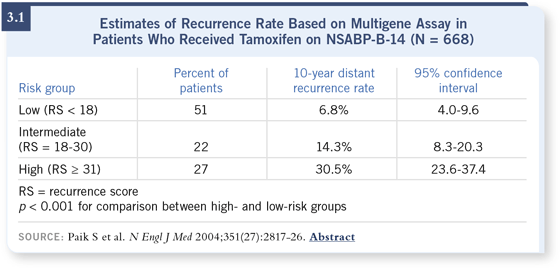
The next step was to validate the index prospectively in another data set. For that data set, we chose to evaluate the 668 tamoxifen-treated patients from NSABP-B-14. The goal was to see whether the recurrence score would separate patients at lower risk from those at higher risk for recurrence (Paik 2004).
 DR LOVE: What specifically was seen when you looked at the tamoxifen arm of the NSABP-B-14 study in terms of the recurrence score? DR LOVE: What specifically was seen when you looked at the tamoxifen arm of the NSABP-B-14 study in terms of the recurrence score?
 DR MAMOUNAS: The recurrence score can range from zero to 100. We found that patients with a recurrence score of less than 18 had a 10-year distant recurrence rate of about 6.8 percent, with very narrow confidence intervals. Patients with a high recurrence score (31 or greater) had about a 30.5 percent 10-year distant recurrence rate. Patients with a recurrence score that fell in between 18 and 31 had an intermediate risk of 10-year recurrence, which was about 15 percent (Paik 2004a; [3.1]). DR MAMOUNAS: The recurrence score can range from zero to 100. We found that patients with a recurrence score of less than 18 had a 10-year distant recurrence rate of about 6.8 percent, with very narrow confidence intervals. Patients with a high recurrence score (31 or greater) had about a 30.5 percent 10-year distant recurrence rate. Patients with a recurrence score that fell in between 18 and 31 had an intermediate risk of 10-year recurrence, which was about 15 percent (Paik 2004a; [3.1]).
 DR MAMOUNAS: The next step was to see whether the recurrence score went above and beyond prognosis; maybe it would provide a prediction of response to therapy. There was good reason to look at that, because the recurrence score index contains genes that have traditionally been associated with response to therapy. DR MAMOUNAS: The next step was to see whether the recurrence score went above and beyond prognosis; maybe it would provide a prediction of response to therapy. There was good reason to look at that, because the recurrence score index contains genes that have traditionally been associated with response to therapy.
For example, low ER positivity versus high ER positivity: We know from neoadjuvant chemotherapy studies that ER negativity has been associated with higher rates of pathologic complete response. Studies have shown that high proliferation and poor nuclear grade are factors associated with chemotherapy response. Therefore, we set out to assess the benefit from adjuvant tamoxifen and adjuvant chemotherapy according to the recurrence score.
We did that with the two studies we used to develop and validate the recurrence score. First, we looked at the NSABP-B-14 study. We ended up having approximately 645 patients for whom we had tissue blocks and who were randomly assigned to tamoxifen or placebo. The idea was to see whether the benefit from tamoxifen would be seen in patients with low, intermediate and high recurrence scores or whether there would be a differential benefit from chemotherapy in these three groups.
Patients with a low recurrence score and those with an intermediate recurrence score benefit significantly from adjuvant tamoxifen. Patients with a high recurrence score seem to have no benefit from adjuvant tamoxifen (Paik 2004b). Now you can take these data with a grain of salt, because there is some uncertainty as the numbers are relatively small, and the threshold for using hormonal therapy is much lower than the threshold for using chemotherapy. I haven’t changed my practice to avoid using hormonal therapy in patients with a high recurrence score.
 DR LOVE: We should also clarify that this data set was looking at both premenopausal and postmenopausal patients. DR LOVE: We should also clarify that this data set was looking at both premenopausal and postmenopausal patients.
 DR MAMOUNAS: Exactly. And, of course, it was looking at tamoxifen. One can start questioning, based on this data, why patients have a high recurrence score. Well, they usually have low PR, high HER2, high nuclear grade. Therefore, these may be the patients who benefit from the aromatase inhibitors if they are postmenopausal. I’m not willing to give up hormonal therapy for these patients, but I would certainly think, at least for postmenopausal patients, it would be a good group to be more biased towards the aromatase inhibitors over tamoxifen. DR MAMOUNAS: Exactly. And, of course, it was looking at tamoxifen. One can start questioning, based on this data, why patients have a high recurrence score. Well, they usually have low PR, high HER2, high nuclear grade. Therefore, these may be the patients who benefit from the aromatase inhibitors if they are postmenopausal. I’m not willing to give up hormonal therapy for these patients, but I would certainly think, at least for postmenopausal patients, it would be a good group to be more biased towards the aromatase inhibitors over tamoxifen.
 Track 5 Track 5
 DR LOVE: In December 2004, Dr Paik presented the second data set in this project. Can you review that? DR LOVE: In December 2004, Dr Paik presented the second data set in this project. Can you review that? |
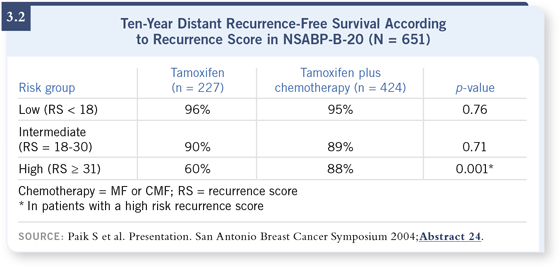
 DR MAMOUNAS: That was the more important finding. We utilized a study the NSABP conducted following NSABP-B-14 — it was NSABP-B-20, which compared tamoxifen alone to tamoxifen plus one of two chemotherapy regimens, either methotrexate and 5-FU (MF) or CMF, in patients with node-negative, ER-positive disease. For all practical purposes, both chemotherapy regimens performed equally well and better than tamoxifen alone. So the overall trial had shown about a 30 percent reduction in risk of recurrence (Fisher 1997). DR MAMOUNAS: That was the more important finding. We utilized a study the NSABP conducted following NSABP-B-14 — it was NSABP-B-20, which compared tamoxifen alone to tamoxifen plus one of two chemotherapy regimens, either methotrexate and 5-FU (MF) or CMF, in patients with node-negative, ER-positive disease. For all practical purposes, both chemotherapy regimens performed equally well and better than tamoxifen alone. So the overall trial had shown about a 30 percent reduction in risk of recurrence (Fisher 1997).
We looked at the benefit of adjuvant chemotherapy according to the recurrence score. It turns out that patients with a low recurrence score received no benefit from chemotherapy. In fact, at 10 years, the distant disease-free survival rate was 96 percent for patients on tamoxifen alone and 95 percent for patients on tamoxifen plus chemotherapy.
Patients with an intermediate recurrence score also did not seem to have much benefit. The 10-year distant recurrence-free survival was approximately 90 percent for both patients treated with tamoxifen alone and those treated with tamoxifen plus chemotherapy (Paik 2004b; [3.2]). However, in that group of patients, the confidence intervals around the estimates were somewhat wide, so we could not exclude some benefit. In fact, the odds ratio was about 0.6, so it could be up to a 40 percent reduction.
What was very interesting was that the benefit was seen in the patients with a high recurrence score. In those patients, the absolute improvement in distant disease-free survival with chemotherapy was 28 percent, or a 75 percent relative reduction in the odds of recurrence. The group that received tamoxifen alone, at 10 years, had a 60 percent distant disease-free survival, and it was 88 percent when they received tamoxifen plus chemotherapy (Paik 2004b; [3.2]).
 DR LOVE: Those numbers were shocking and, to many people, unexpected. DR LOVE: Those numbers were shocking and, to many people, unexpected.
 DR MAMOUNAS: We’ve never seen such differences in any subset of patients with breast cancer. I like to quote what George Sledge said when he saw these data. He said, “This makes CMF look like a targeted regimen.” In fact, that’s true. In other words, we found a signature that predicts a huge benefit from a regimen that otherwise was almost ready to become obsolete. DR MAMOUNAS: We’ve never seen such differences in any subset of patients with breast cancer. I like to quote what George Sledge said when he saw these data. He said, “This makes CMF look like a targeted regimen.” In fact, that’s true. In other words, we found a signature that predicts a huge benefit from a regimen that otherwise was almost ready to become obsolete.
 Track 8 Track 8
 DR LOVE: Would you summarize the NSABP-B-32 sentinel node trial? DR LOVE: Would you summarize the NSABP-B-32 sentinel node trial? |
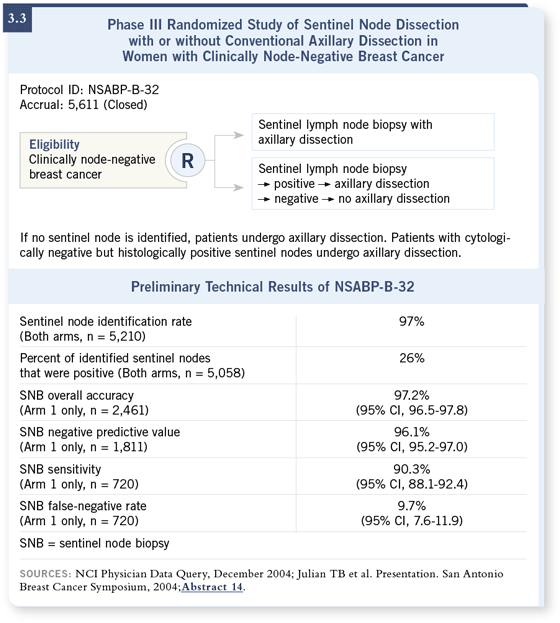
 DR MAMOUNAS: NSABP-B-32 was a large randomized trial comparing sentinel node biopsy followed by standard axillary dissection to sentinel node biopsy alone, provided the sentinel node was negative intraoperatively or postoperatively. This was the largest randomized trial of sentinel node biopsy, with over 5,600 patients ( Julian 2004; [3.3]). DR MAMOUNAS: NSABP-B-32 was a large randomized trial comparing sentinel node biopsy followed by standard axillary dissection to sentinel node biopsy alone, provided the sentinel node was negative intraoperatively or postoperatively. This was the largest randomized trial of sentinel node biopsy, with over 5,600 patients ( Julian 2004; [3.3]).
If the sentinel node were positive, then for both groups, an axillary dissection would take place. This was a study for patients with sentinel node-negative disease to evaluate complete axillary dissection. We presented a technical report comparing the identification rates for the sentinel node and false-negative rates. What we found was that the identification rate was about 97 percent, and it became better as surgeons performed more biopsies ( Julian 2004; [3.3]).
The false-negative rate was about 9.7 percent ( Julian 2004; [3.3]). Interestingly enough, false-negative rates did not seem to improve that much, or not significantly, with time and the more procedures that the surgeons performed. The false-negative rates appear to be inherent to the sentinel node biopsy procedure.
 Track 10 Track 10
 DR LOVE: Can you summarize where we are right now with the adjuvant aromatase inhibitors in postmenopausal women — up front and after two, three years or after five years of adjuvant tamoxifen? DR LOVE: Can you summarize where we are right now with the adjuvant aromatase inhibitors in postmenopausal women — up front and after two, three years or after five years of adjuvant tamoxifen? |
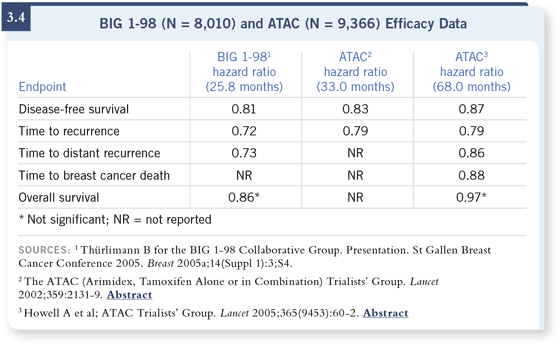
 DR MAMOUNAS: There are a total of six randomized trials that have reported benefits with aromatase inhibitors either above and beyond tamoxifen or as extended adjuvant therapy. The interesting pattern we are now seeing is that if we compare different aromatase inhibitors in the same setting, the results are pretty much consistent. We now have data looking at letrozole as up-front adjuvant therapy with a relative reduction in recurrence of about 19 percent (Thürlimann 2005; [3.4]), which was very similar to what was seen with the ATAC trial and anastrozole (Howell 2005; [3.4]). DR MAMOUNAS: There are a total of six randomized trials that have reported benefits with aromatase inhibitors either above and beyond tamoxifen or as extended adjuvant therapy. The interesting pattern we are now seeing is that if we compare different aromatase inhibitors in the same setting, the results are pretty much consistent. We now have data looking at letrozole as up-front adjuvant therapy with a relative reduction in recurrence of about 19 percent (Thürlimann 2005; [3.4]), which was very similar to what was seen with the ATAC trial and anastrozole (Howell 2005; [3.4]).
There are three studies in which the aromatase inhibitors are introduced after two to three years of adjuvant tamoxifen, showing recurrence reductions in the range of about 30 to 40 percent. The ITA trial was a smaller study with 448 patients and about a 60 percent reduction with anastrozole (Boccardo 2005). The Intergroup Exemestane Study (IES) (Coombes 2004), as well as the ARNO/ABCSG trials ( Jakesz 2005) have shown reductions in the range of 30 to 40 percent with anastrozole or exemestane.
Of course, we have a third setting, that of extended adjuvant therapy, in which letrozole also produced about a 40 percent reduction compared to placebo after five years of adjuvant tamoxifen (Goss 2005). Based on these results, clearly, aromatase inhibitors have entered the adjuvant therapy setting.
The bigger question nowadays remains: What is the best setting in which to introduce the aromatase inhibitors — up front or after two to three years or after five years of tamoxifen? Most oncologists and surgeons will switch a patient who is on two to three years of tamoxifen — and not necessarily wait the five years — or prescribe an aromatase inhibitor up front.
 Track 11 Track 11
 DR LOVE: What is the future direction of the NSABP in terms of the next generation of adjuvant endocrine therapy trials? DR LOVE: What is the future direction of the NSABP in terms of the next generation of adjuvant endocrine therapy trials? |
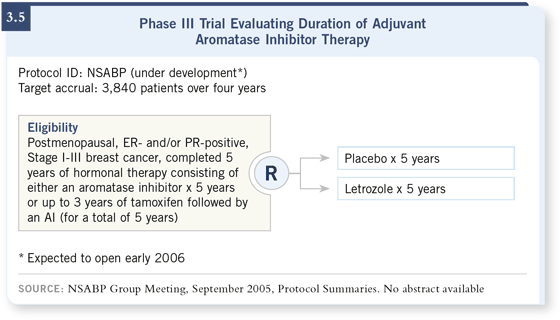
 DR MAMOUNAS: We believe that this is an important time to study the question of duration of aromatase inhibitor therapy. So the NSABP has designed a study to take patients who complete five years of an aromatase inhibitor — either anastrozole, letrozole or exemestane — or patients who complete five years of hormonal therapy that consists of at least two to three years of an aromatase inhibitor and randomly assign them to an aromatase inhibitor — in this case, letrozole — versus placebo (3.5). DR MAMOUNAS: We believe that this is an important time to study the question of duration of aromatase inhibitor therapy. So the NSABP has designed a study to take patients who complete five years of an aromatase inhibitor — either anastrozole, letrozole or exemestane — or patients who complete five years of hormonal therapy that consists of at least two to three years of an aromatase inhibitor and randomly assign them to an aromatase inhibitor — in this case, letrozole — versus placebo (3.5).
Essentially, we are repeating what was done in the NSABP-B-14 trial with tamoxifen, but now with aromatase inhibitors. I believe that this question should be studied prospectively, and the existing databases or continuation of current trials will not provide a definitive answer. We are planning on continuing the aromatase inhibitor therapy for five years.
Select publicaitons
|

