You are here: Home:
BCU 3 | 2006: Adjuvant Endocrine Therapy for Patients with ER- and/or
PR-Positive Disease

| Tracks 1-7 |
| Track 1 |
Clinical trials evaluating aromatase inhibitors in the neoadjuvant setting |
| Track 2 |
Overview of aromatase inhibitors trials |
| Track 3 |
Aromatase inhibitors versus tamoxifen |
| Track 4 |
Bone effects with aromatase inhibitors |
|
| Track 5 |
Gynecologic side effects with aromatase inhibitors and tamoxifen |
| Track 6 |
Time course of recurrence of ER/PR-positive disease |
| Track 7 |
Extended adjuvant therapy with aromatase inhibitors |
|
|
Select Excerpts from the Interview
 Track 2
Track 2
 DR LOVE: Richard, can you summarize the data from the trials of
adjuvant aromatase inhibitors? DR LOVE: Richard, can you summarize the data from the trials of
adjuvant aromatase inhibitors? |
 DR SAINSBURY: ATAC (Howell 2005) and BIG 1-98 (Thürlimann 2005)
studied a population starting afresh. IES (Coombes 2004), ARNO 95 and
ABCSG-8 (Jakesz 2005a) enrolled patients who had already received two or
three years of adjuvant tamoxifen, and MA17 (Goss 2005) enrolled patients
who completed five years of adjuvant tamoxifen.
DR SAINSBURY: ATAC (Howell 2005) and BIG 1-98 (Thürlimann 2005)
studied a population starting afresh. IES (Coombes 2004), ARNO 95 and
ABCSG-8 (Jakesz 2005a) enrolled patients who had already received two or
three years of adjuvant tamoxifen, and MA17 (Goss 2005) enrolled patients
who completed five years of adjuvant tamoxifen.
So for some of these studies, you’ve already eliminated patients who had
earlier relapses. Therefore, the studies are not strictly comparable and have to
be considered in context.
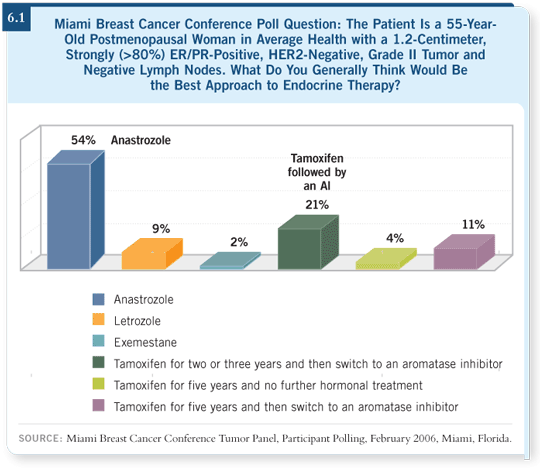
ATAC (Howell 2005) and BIG 1-98 (Thürlimann 2005) both showed that
up-front anastrozole and letrozole, respectively, were better than tamoxifen
for disease-free survival. The ATAC study showed an apparent blunting of
the early relapses, suggesting that to avoid recurrences you need to start the
aromatase inhibitor early and not switch at two to three years.
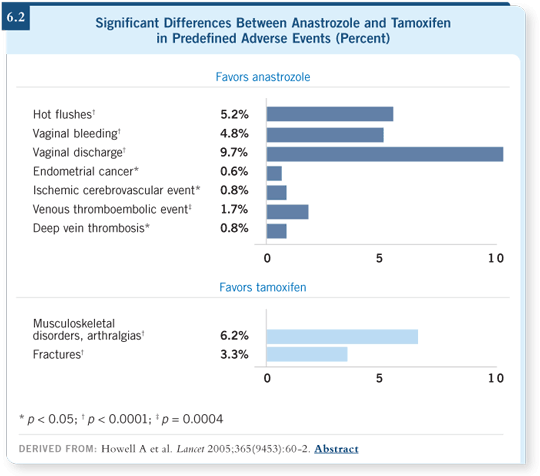
We’re obtaining increasing evidence that avoiding recurrences will affect
mortality down the line. Both studies demonstrated different side effects, with
generally reduced toxicities for the aromatase inhibitors compared to tamoxifen. Worry arose about the impact on bone, which is a manageable concern
but something we still need to watch carefully (Howell 2005; Thürlimann
2005; [6.2]). Anastrozole has a slightly longer follow-up.
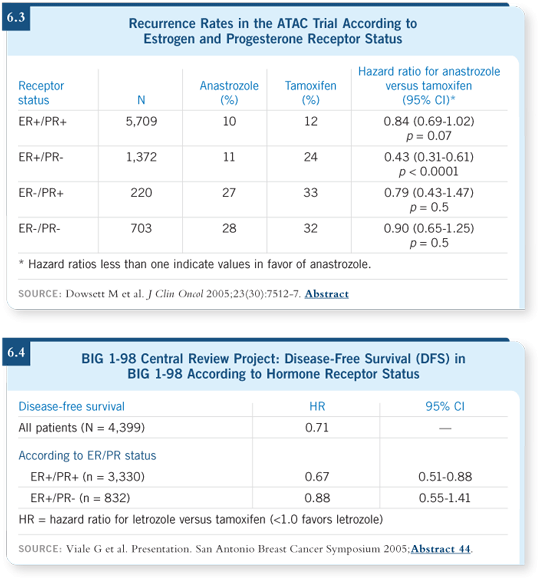
A retrospective analysis of ATAC indicated an apparent benefit for the subset
with ER-positive, PR-negative disease (Dowsett 2005; [6.3]), which was not
found in BIG 1-98, even when the tumors were reassessed centrally (Viale
2005; [6.4]). That is probably a fluke of subset analyses.
In the switching trials, the aromatase inhibitors are better than tamoxifen for
relapse-free survival and have better side-effect profiles (Coombes 2004; Jakesz
2005a; Boccardo 2005). The combined analysis of ABCSG-8, ARNO 95 and
the Italian study (ITA) demonstrated an overall survival benefit for anastrozole ( Jonat 2005; [6.5]); however, those studies were not intended to be combined,
so I believe that survival benefit is artifactual.

 Track 3
Track 3
 DR LOVE: George, what are your thoughts about the lack of a clear
survival benefit in the trials of adjuvant aromatase inhibitors? DR LOVE: George, what are your thoughts about the lack of a clear
survival benefit in the trials of adjuvant aromatase inhibitors? |
 DR SLEDGE: I’ve not been as worried as others. In recent years, we’ve been
fooled into thinking we should see survival advantages two or three years out
on adjuvant trials. If one looks at the adjuvant tamoxifen trials from the 1980s,
one sees that it was common for us to wait five, six or seven years to see a
survival advantage.
DR SLEDGE: I’ve not been as worried as others. In recent years, we’ve been
fooled into thinking we should see survival advantages two or three years out
on adjuvant trials. If one looks at the adjuvant tamoxifen trials from the 1980s,
one sees that it was common for us to wait five, six or seven years to see a
survival advantage.
So it doesn’t at all surprise me that a lot of our trials haven’t yet shown those
advantages. I think they’re going to emerge. If you significantly reduce a
woman’s likelihood of relapsing in her liver, lungs and bones, it will translate
into a survival advantage unless these agents are significantly more toxic in
some nonbreast cancer fashion.
 DR LOVE: Peter, if the efficacy of the aromatase inhibitors were identical to
tamoxifen, would they still be preferable because of the side-effect and toxicity
profiles?
DR LOVE: Peter, if the efficacy of the aromatase inhibitors were identical to
tamoxifen, would they still be preferable because of the side-effect and toxicity
profiles?
 DR RAVDIN: Yes. For postmenopausal women in their sixties, the risk of
cancer and the risk of thrombotic events are both very significant. I think if
it were the other way around and we had the aromatase inhibitors first and
we were trying to introduce tamoxifen, tamoxifen might not make the bar,
except for premenopausal patients. I think toxicity is a major reason to select
an aromatase inhibitor over tamoxifen.
DR RAVDIN: Yes. For postmenopausal women in their sixties, the risk of
cancer and the risk of thrombotic events are both very significant. I think if
it were the other way around and we had the aromatase inhibitors first and
we were trying to introduce tamoxifen, tamoxifen might not make the bar,
except for premenopausal patients. I think toxicity is a major reason to select
an aromatase inhibitor over tamoxifen.
 Track 5
Track 5
 DR LOVE: Richard, can you comment on the gynecologic data from the ATAC trial that were presented at the 2005 San Antonio Breast Cancer Symposium (Duffy 2005)? DR LOVE: Richard, can you comment on the gynecologic data from the ATAC trial that were presented at the 2005 San Antonio Breast Cancer Symposium (Duffy 2005)? |
 DR SAINSBURY: In the ATAC trial, the hysterectomy rate went down from six percent with tamoxifen to approximately two percent with anastrozole.
That’s a major health issue. Any woman on tamoxifen who has a bleed is investigated, which is expensive and invasive (Duffy 2005; [6.5]). Sean Duffy conducted the prospective endometrial study (Duffy 2006). In fact, we’ve gained a lot of information about what’s happening in the uteri of these women that we didn’t know about before. It’s clear that the aromatase inhibitors
are much less toxic to the uterus than tamoxifen is.
DR SAINSBURY: In the ATAC trial, the hysterectomy rate went down from six percent with tamoxifen to approximately two percent with anastrozole.
That’s a major health issue. Any woman on tamoxifen who has a bleed is investigated, which is expensive and invasive (Duffy 2005; [6.5]). Sean Duffy conducted the prospective endometrial study (Duffy 2006). In fact, we’ve gained a lot of information about what’s happening in the uteri of these women that we didn’t know about before. It’s clear that the aromatase inhibitors
are much less toxic to the uterus than tamoxifen is.
Select publications

