
| Tracks 1-6 |
| Track 1 |
Historical development of adjuvant chemotherapy |
| Track 2 |
ECOG-E1199 adjuvant trial: Evaluating type and schedule of taxanes |
| Track 3 |
GEICAM 9906: Adjuvant FE90C
versus FE90C followed by weekly paclitaxel |
|
| Track 4 |
US Oncology adjuvant trial comparing AC to TC |
| Track 5 |
Anthracycline- versus nonanthra-
cycline-based adjuvant chemotherapy |
| Track 6 |
Prediction of response to adjuvant chemotherapy |
|
|
Select Excerpts from the Interview
 Track 2
Track 2
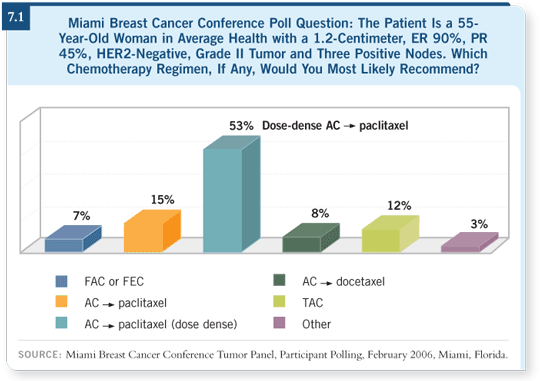
 DR LOVE: Would you provide an overview of the adjuvant ECOG-E1199
trial that was presented at the 2005 San Antonio meeting? DR LOVE: Would you provide an overview of the adjuvant ECOG-E1199
trial that was presented at the 2005 San Antonio meeting? |
 DR RAVDIN: The ECOG-E1199 trial asked two questions: Which taxane
and which schedule (Sparano 2005) are optimal as adjuvant therapy? Patients received AC, and then the standard arm was paclitaxel every three weeks for
four cycles. Another arm was a substitution of docetaxel for paclitaxel, and
two arms evaluated these agents in weekly regimens.
DR RAVDIN: The ECOG-E1199 trial asked two questions: Which taxane
and which schedule (Sparano 2005) are optimal as adjuvant therapy? Patients received AC, and then the standard arm was paclitaxel every three weeks for
four cycles. Another arm was a substitution of docetaxel for paclitaxel, and
two arms evaluated these agents in weekly regimens.
If you look at paclitaxel versus docetaxel, you see no superiority in a two-by-two comparison between the two agents. If you look at every three weeks
versus weekly, you see no difference in efficacy.
However, the devil is in the details, and as clinicians we all want to know the
one-by-four comparisons. The results are consistent with what we’ve seen in
metastatic disease.
The weekly paclitaxel regimen was the best, with almost a 20 percent better
hazard ratio than the standard arm (7.2). Docetaxel, given every three weeks,
also looked somewhat better.
In both of those cases, however, the difference was a trend and was not statistically significant. The weekly paclitaxel arm looked best in terms of overall
survival, but this is a very early analysis not dignified by p-values.
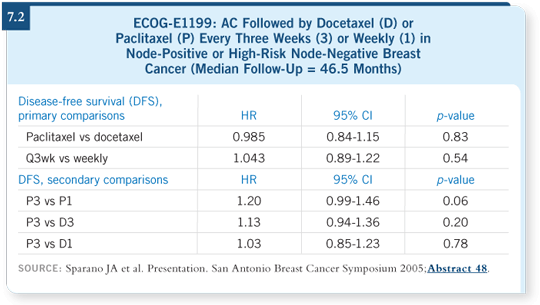
What about toxicity? The weekly paclitaxel arm seemed to provide additional
benefit without additional risk of febrile neutropenia, whereas the docetaxel
arm was associated with additional febrile neutropenia (7.3).
A conclusion from this study has to be that weekly paclitaxel in adjuvant
therapy appears promising, and the hazard ratios for the weekly arm of E1199
looked very similar to those of dose-dense therapy.
 Track 4
Track 4
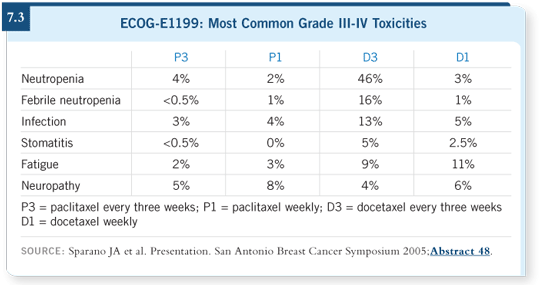
 DR LOVE: Could you briefly review the results of the US Oncology
adjuvant trial comparing AC to docetaxel/cyclophosphamide (TC)? DR LOVE: Could you briefly review the results of the US Oncology
adjuvant trial comparing AC to docetaxel/cyclophosphamide (TC)? |
 DR RAVDIN: A striking improvement in disease-free survival and a large
improvement in overall survival have been seen with TC in this study.
DR RAVDIN: A striking improvement in disease-free survival and a large
improvement in overall survival have been seen with TC in this study.
These improvements were obtained without a major difference in toxicity.
A very slight increase occurs in neutropenia and fever rate with TC, but
other advantages appear in the taxane-containing arm, with less nausea and
vomiting (Jones 2005; [7.4]).
No major difference is seen between ER-positive and ER-negative disease in
terms of the superiority of the taxane arm. A promising area in the development of adjuvant chemotherapy is that we may be able to substitute for the use
of anthracyclines in adjuvant therapy, particularly in special populations, such
as those who will be receiving trastuzumab.
 Track 5
Track 5
 DR LOVE: Joyce, can you comment on the study of adjuvant AC versus
docetaxel/cyclophosphamide (TC) (Jones 2005)? We know from our
Patterns of Care study that right now AC is the most common regimen
used by oncologists for patients with node-negative disease. DR LOVE: Joyce, can you comment on the study of adjuvant AC versus
docetaxel/cyclophosphamide (TC) (Jones 2005)? We know from our
Patterns of Care study that right now AC is the most common regimen
used by oncologists for patients with node-negative disease. |
 DR O’SHAUGHNESSY: TC is definitely a better-tolerated regimen than AC.
While it was not reported, any physician who took care of the patients on
both arms is aware that less fatigue occurred with the TC because docetaxel
at 75 mg/m2 is not particularly fatiguing. With AC, you can get that kind of
prolonged queasiness, and for some patients it brings them down for a week
or so.
DR O’SHAUGHNESSY: TC is definitely a better-tolerated regimen than AC.
While it was not reported, any physician who took care of the patients on
both arms is aware that less fatigue occurred with the TC because docetaxel
at 75 mg/m2 is not particularly fatiguing. With AC, you can get that kind of
prolonged queasiness, and for some patients it brings them down for a week
or so.
TC is much less nauseating and much better tolerated. It’s a night and day
difference, in my opinion. I have stopped using AC now for patients for whom
I was using it. Now I use TC because of the six percent absolute improvement
in disease-free survival.
Select publications

