
| Tracks 1-19 |
| Track 1 |
Introduction |
| Track 2 |
Ovarian suppression concurrent with adjuvant chemotherapy to preserve ovarian functioning |
| Track 3 |
SWOG trial of chemotherapy with or without ovarian suppression in patients with ER-negative disease |
| Track 4 |
Potential rationale for the failure of ovarian suppression during chemotherapy to preserve fertility |
| Track 5 |
Natural history of ovarian functioning |
| Track 6 |
Ovarian stimulation, ova recovery, fertilization and cryopreservation to preserve fertility |
| Track 7 |
Selection of adjuvant chemotherapy for women who want to preserve fertility |
| Track 8 |
Methotrexate/fluorouracil as a therapeutic option for avoiding amenorrhea |
| Track 9 |
Effect of pregnancy on risk of recurrence in patients with previously treated breast cancer |
| Track 10 |
Importance of barrier contraception
while receiving systemic therapy |
|
| Track 11 |
Impact of tamoxifen and chemotherapy on fetal outcomes during pregnancy |
| Track 12 |
Safety to a fetus of sentinel lymph node biopsy |
| Track 13 |
Clinical trials of adjuvant hormonal therapy in
premenopausal patients |
| Track 14 |
Utility of ovarian suppression in combination with chemotherapy and tamoxifen |
| Track 15 |
Implications of data from
adjuvant aromatase inhibitor
trials on clinical practice |
| Track 16 |
Aromatase inhibitors in women with chemotherapy-induced amenorrhea |
| Track 17 |
Future clinical research questions in patients with ER/PR-positive tumors |
| Track 18 |
Adjuvant trastuzumab monotherapy |
| Track 19 |
Use of fulvestrant in clinical practice |
|
|
Select Excerpts from the Interview
 Track 5
Track 5
 DR LOVE: Can you talk about what we know about ovarian function and resumption of menses in premenopausal women after chemotherapy? DR LOVE: Can you talk about what we know about ovarian function and resumption of menses in premenopausal women after chemotherapy? |
 DR FOX: A great deal of data have been collected through NSABP trial B-30 and a project that was initiated by the late Dr Jeanne Petrek, which examined the natural history of loss of menstrual function during breast cancer treatment.
DR FOX: A great deal of data have been collected through NSABP trial B-30 and a project that was initiated by the late Dr Jeanne Petrek, which examined the natural history of loss of menstrual function during breast cancer treatment.
In this first large-scale evaluation of the effect of taxane-based chemotherapy on menstrual function, it was observed that the likelihood of becoming amenorrheic was age related, which you would expect with any chemotherapy regimen.
In a nonrandomized setting, in terms of amenorrhea, those patients who received taxanes fared a little worse than those who had not received taxanes (Petrek 2005).
These data also demonstrated that if a premenopausal woman is treated with chemotherapy and becomes amenorrheic, it is inappropriate to assume that she will remain in a state of real menopause. Based on the natural history data, it appears to take two years to establish with some certainty that a woman will remain in a state of menopause.
If a 45-year-old woman — five years from the mean age of menopause — receives chemotherapy and becomes amenorrheic, I do not believe we can be assured that her ovaries will remain in a state of menopause until we’ve followed her for two years.
 Track 7
Track 7
 DR LOVE: How do you present the option of chemotherapy to premenopausal
women who may be willing to accept an increased risk of relapse to maintain fertility? DR LOVE: How do you present the option of chemotherapy to premenopausal
women who may be willing to accept an increased risk of relapse to maintain fertility? |
 DR FOX: The friendliest regimen with respect to not inducing permanent amenorrhea appears to be four cycles of AC (Petrek 2006). This regimen has been around long enough that we have accumulated experience, and in aggregate,
AC produces less amenorrhea than CMF. Based on ASCO presentations, evidence suggests that the addition of a taxane may increase the amenorrhea rates more than AC alone.
DR FOX: The friendliest regimen with respect to not inducing permanent amenorrhea appears to be four cycles of AC (Petrek 2006). This regimen has been around long enough that we have accumulated experience, and in aggregate,
AC produces less amenorrhea than CMF. Based on ASCO presentations, evidence suggests that the addition of a taxane may increase the amenorrhea rates more than AC alone.
 Track 13
Track 13
 DR LOVE: Can you provide an update of clinical research on adjuvant hormonal therapy for premenopausal patients? DR LOVE: Can you provide an update of clinical research on adjuvant hormonal therapy for premenopausal patients? |
 DR FOX: The most significant challenge in developing new therapeutic strategies
for premenopausal women with hormone receptor-positive breast cancers is the issue of ovarian suppression.
DR FOX: The most significant challenge in developing new therapeutic strategies
for premenopausal women with hormone receptor-positive breast cancers is the issue of ovarian suppression.
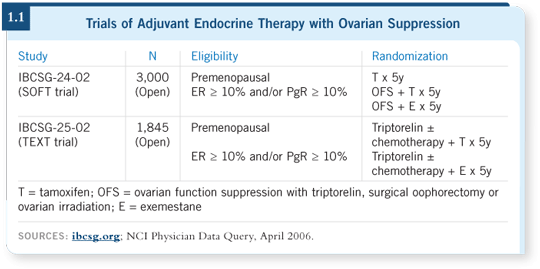
We are participating in one of the two largest clinical trials (1.1) addressing this issue: the SOFT trial, which randomly assigns premenopausal women with receptor-positive cancers to receive tamoxifen alone, ovarian suppression for five years with tamoxifen or ovarian suppression for five years with exemestane.
 Track 15
Track 15
 DR LOVE: What are your thoughts about the clinical trials of aromatase
inhibitors for postmenopausal patients and their current implications for
clinical practice? DR LOVE: What are your thoughts about the clinical trials of aromatase
inhibitors for postmenopausal patients and their current implications for
clinical practice? |
 DR FOX: At the moment, you have to evaluate the data based on where the
patient is in her course of treatment.
DR FOX: At the moment, you have to evaluate the data based on where the
patient is in her course of treatment.
The ATAC trial addresses one scenario, which is treatment of the newly
diagnosed postmenopausal patient with estrogen receptor-positive breast
cancer, and this trial provides irrefutable evidence that anastrozole is superior
to tamoxifen with respect to reducing the risk of recurrence. The available
data suggest that five years of an aromatase inhibitor is the best therapy for the
newly diagnosed patient (Howell 2005).
The second scenario is that in which the patient is in the middle of a course
of therapy. The International Exemestane Study and the trials of anastrozole,
which were similarly constructed (Coombes 2005; Boccardo 2005; Jakesz
2005), were designed to enroll patients at the midpoint of a course of tamoxifen therapy and measure outcomes from the point of changing treatment.
Patients were randomly assigned to continue tamoxifen or to switch to an
aromatase inhibitor for the balance of the five-year period.
For the patient in the middle of a course of tamoxifen therapy or the
premenopausal woman who’s become amenorrheic from chemotherapy and has
been on tamoxifen and amenorrheic for two years, it is appropriate to switch
to an aromatase inhibitor.
A third situation is that in which the patient has completed five years of
tamoxifen. The only trial addressing this is MA17, which demonstrates that
letrozole produces a small but measurable reduction in the risk of recurrence
and an indication of a survival benefit in women with node-positive disease
(Goss 2005).
 Track 16
Track 16
 DR LOVE: Do you measure FSH, LH and estradiol levels in a premenopausal
woman who becomes amenorrheic on chemotherapy before administering
an aromatase inhibitor? DR LOVE: Do you measure FSH, LH and estradiol levels in a premenopausal
woman who becomes amenorrheic on chemotherapy before administering
an aromatase inhibitor? |
 DR FOX: If our patients report two years of amenorrhea and we are considering
switching them to an aromatase inhibitor, we always try to corroborate
that information with an estradiol and an FSH level, recognizing the occasional shortcomings of either of those measurements.
DR FOX: If our patients report two years of amenorrhea and we are considering
switching them to an aromatase inhibitor, we always try to corroborate
that information with an estradiol and an FSH level, recognizing the occasional shortcomings of either of those measurements.
In my own practice, I require that a patient have nonmeasurable levels of estrogen and an elevated FSH in the postmenopausal range before prescribing an aromatase inhibitor.
Select publications

