
| Tracks 1-19 |
| Track 1 |
Introduction |
| Track 2 |
Clinical trial results with
nanoparticle albumin-bound (nab) paclitaxel |
| Track 3 |
Clinical use of single-agent nab paclitaxel for metastatic disease |
| Track 4 |
Comparison of neurotoxicity between paclitaxel and nab paclitaxel |
| Track 5 |
Predictors of response to nab paclitaxel |
| Track 6 |
Optimal first-line taxane therapy |
| Track 7 |
US Oncology trial results comparing adjuvant AC versus TC (docetaxel/cyclophosphamide) |
| Track 8 |
ECOG-E1199: Evaluating type and schedule of taxanes |
| Track 9 |
ECOG-E2100: Paclitaxel with
or without bevacizumab as
first-line therapy for metastatic disease |
| Track 10 |
Continuation of bevacizumab upon disease progression |
|
| Track 11 |
Impact of cost and reimbursement on the use of
novel therapeutics |
| Track 12 |
First-line therapy for patients with minimally symptomatic disease |
| Track 13 |
Selection of first-line therapy for chemotherapy-naïve patients |
| Track 14 |
First-line therapy for patients progressing after adjuvant taxane therapy |
| Track 15 |
Use of capecitabine in the first-line metastatic setting |
| Track 16 |
EFECT: Evaluation of Fulvestrant and Exemestane Clinical Trial |
| Track 17 |
SoFEA: Study of Fulvestrant with or without concomitant Exemestane versus Anastrozole |
| Track 18 |
Aromatase inhibitors plus an LHRH agonist for premenopausal patients with metastatic disease |
| Track 19 |
High-dose fulvestrant in premenopausal patients |
|
|
Select Excerpts from the Interview
 Tracks 2, 4
Tracks 2, 4
 DR LOVE: What is the current status of nanoparticle albumin-bound (nab) paclitaxel in the treatment of metastatic breast cancer, and how does it compare with paclitaxel in terms of neurotoxicity and efficacy? DR LOVE: What is the current status of nanoparticle albumin-bound (nab) paclitaxel in the treatment of metastatic breast cancer, and how does it compare with paclitaxel in terms of neurotoxicity and efficacy? |
 DR GRADISHAR: In the last year, the use of nab paclitaxel has increased significantly, and as clinicians gain more experience with it, they get a better
understanding of when it’s used most effectively.
DR GRADISHAR: In the last year, the use of nab paclitaxel has increased significantly, and as clinicians gain more experience with it, they get a better
understanding of when it’s used most effectively.
When nab paclitaxel was developed, an underlying notion was that if you
eliminated the solvents, all the neuropathy would disappear.
It has been demonstrated that Cremophor® is significantly related to the
development of peripheral neurotoxicity that is long-lived and potentially not
completely reversible once it develops in patients.
In the pivotal trial, an every three-week schedule of nab paclitaxel at 260
mg/m2 was compared to the standard dose of paclitaxel at 175 mg/m2. Interestingly, the rate of Grade III neuropathy with nab paclitaxel was in the range
of 10 percent compared to two percent for the patients who received paclitaxel
(Gradishar 2005).
However, what appears to be consistent with nab paclitaxel in both the every
three-week and weekly schedules is that the neuropathy seems to be different
than that seen with paclitaxel.
With nab paclitaxel it appears to be more short-lived, with the majority of
patients being able to resume therapy within three weeks.
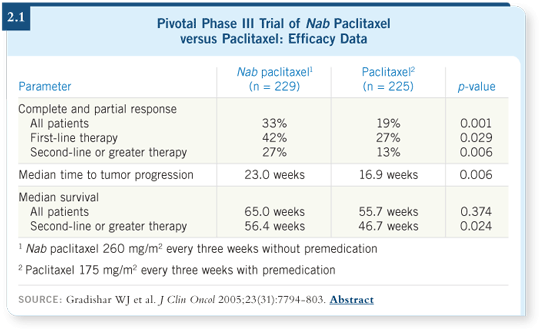
In terms of efficacy, approximately 40 percent of the patients had not received
prior therapy for metastatic disease, and in that group of patients, the response
rate for nab paclitaxel was far superior to the response rate among patients
treated with paclitaxel (Gradishar 2005; [2.1]).
A CALGB trial will be evaluating weekly and every three-week schedules of nab paclitaxel versus paclitaxel in the metastatic disease setting, but we don’t
have any data from that trial yet.
 Track 6
Track 6
 DR LOVE: In your opinion, what is the optimal first-line taxane in the
metastatic setting? DR LOVE: In your opinion, what is the optimal first-line taxane in the
metastatic setting? |
 DR GRADISHAR: The data are still more abundant with both paclitaxel and
docetaxel than with nab paclitaxel, so for basing a decision on the length of
experience, those agents have been around for a longer time.
DR GRADISHAR: The data are still more abundant with both paclitaxel and
docetaxel than with nab paclitaxel, so for basing a decision on the length of
experience, those agents have been around for a longer time.
However, I see no reason to believe that nab paclitaxel will prove inferior to
those drugs with more data. I believe nab paclitaxel will compare favorably, if
not prove to be superior.
When you examine clinical trials that have evaluated docetaxel or paclitaxel in
similar patient populations with metastatic disease, the indirect evidence shows
the activity of nab paclitaxel to be comparable to docetaxel.
These agents may have similar antitumor effects, so one should consider other
factors, including toxicities, patient convenience and cost.
 DR LOVE: If we determine that nab paclitaxel has the same antitumor
effect as docetaxel and paclitaxel, do you believe the advantages of this agent,
in terms of lack of premedication and shorter infusion time, make it the
preferred agent?
DR LOVE: If we determine that nab paclitaxel has the same antitumor
effect as docetaxel and paclitaxel, do you believe the advantages of this agent,
in terms of lack of premedication and shorter infusion time, make it the
preferred agent?
 DR GRADISHAR: That’s an important question. When you think of busy
office practices, the throughput of patients and the convenience to patients are
important. An upside to nab paclitaxel clearly is the shorter infusion time and
the lack of need for premedication.
DR GRADISHAR: That’s an important question. When you think of busy
office practices, the throughput of patients and the convenience to patients are
important. An upside to nab paclitaxel clearly is the shorter infusion time and
the lack of need for premedication.
As for the higher acquisition cost of nab paclitaxel, economic analyses suggest
that some of the downstream expenses related to administering paclitaxel or
docetaxel — specifically the costs of premedications and antibiotics or growth
factors to manage the neutropenias or cytopenias — result in a net savings
with the use of nab paclitaxel.
Although we need more information, I believe we shouldn’t necessarily be put
off by the up-front cost; we should take into account the whole package of
managing the patient’s treatment.

 Track 9
Track 9
 DR LOVE: Would you summarize the clinical trial findings with
bevacizumab in metastatic breast cancer? DR LOVE: Would you summarize the clinical trial findings with
bevacizumab in metastatic breast cancer? |
 DR GRADISHAR: One of the early trials suggested that combining bevacizumab with capecitabine, at least in patients who were heavily pretreated, did
not bring much in the way of additional benefit compared to administering
capecitabine alone (Miller 2005b).
DR GRADISHAR: One of the early trials suggested that combining bevacizumab with capecitabine, at least in patients who were heavily pretreated, did
not bring much in the way of additional benefit compared to administering
capecitabine alone (Miller 2005b).
That was disappointing and in contrast to what has been seen in other disease
sites, particularly colorectal cancer, for which bevacizumab is widely used.
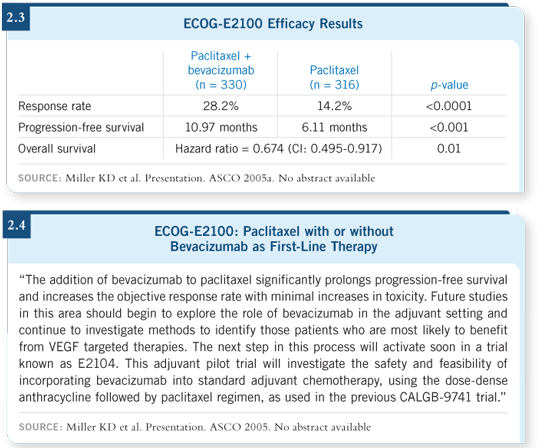
Rather than abandoning the agent in breast cancer, another trial was initiated
comparing paclitaxel with or without bevacizumab. The data clearly showed a
benefit that favored the combination (Miller 2005a; [2.3]).
 DR LOVE: Can you discuss the related XCaliBr study you are chairing with
George Sledge?
DR LOVE: Can you discuss the related XCaliBr study you are chairing with
George Sledge?
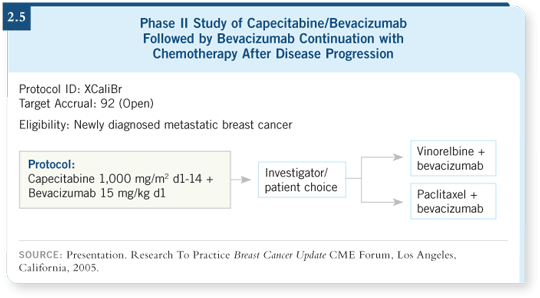
 DR GRADISHAR: Rather than assuming there was no reason to pursue bevacizumab
with a 5-FU-like drug, the XCaliBr trial was designed to evaluate
the combination of bevacizumab and capecitabine as first-line treatment for
metastatic breast cancer (2.5). In this trial, the patients must have HER2-
negative, measurable disease.
DR GRADISHAR: Rather than assuming there was no reason to pursue bevacizumab
with a 5-FU-like drug, the XCaliBr trial was designed to evaluate
the combination of bevacizumab and capecitabine as first-line treatment for
metastatic breast cancer (2.5). In this trial, the patients must have HER2-
negative, measurable disease.
The trial has almost reached its accrual goal, which is around 100 patients,
so we anticipate it will be closed soon. This study will examine the issue of
continuing bevacizumab on disease progression and will try to determine if
there’s any differential effect between bevacizumab with vinorelbine or paclitaxel as second-line therapy.
 Track 15
Track 15
 DR LOVE: Our Patterns of Care studies indicate that clinical investigators
use capecitabine much earlier in the treatment algorithm than physicians
in community practice, and as I recall, you are among the investigators
who use single-agent capecitabine pretty early in metastatic disease. DR LOVE: Our Patterns of Care studies indicate that clinical investigators
use capecitabine much earlier in the treatment algorithm than physicians
in community practice, and as I recall, you are among the investigators
who use single-agent capecitabine pretty early in metastatic disease.
Has that approach changed with the bevacizumab data? |
 DR GRADISHAR: I still believe that capecitabine is a good up-front agent to
use in metastatic disease for many patients, and that hasn’t changed with the
bevacizumab data.
DR GRADISHAR: I still believe that capecitabine is a good up-front agent to
use in metastatic disease for many patients, and that hasn’t changed with the
bevacizumab data.
However, the data that emerge from the XCaliBr study (2.5) may provide
justification for using capecitabine with bevacizumab, assuming the data are
positive and comparable to what we saw in the E2100 study (Miller 2005a).
 DR LOVE: What type of patient do you consider an ideal candidate for front-
line, single-agent capecitabine in the metastatic setting?
DR LOVE: What type of patient do you consider an ideal candidate for front-
line, single-agent capecitabine in the metastatic setting?
 DR GRADISHAR: Capecitabine is comparable to our most active chemotherapy
drugs, but I don’t view any drug as the best agent in a particular situation.
I would use capecitabine for patients with minimal visceral disease such as
small liver metastases, but docetaxel or nab paclitaxel would be fine as well.
DR GRADISHAR: Capecitabine is comparable to our most active chemotherapy
drugs, but I don’t view any drug as the best agent in a particular situation.
I would use capecitabine for patients with minimal visceral disease such as
small liver metastases, but docetaxel or nab paclitaxel would be fine as well.
It’s a judgment call that you make with each patient depending on her
preferences.
Select publications

