
| Tracks 1-11 |
| Track 1 |
Introduction |
| Track 2 |
Overview of adjuvant trastuzumab trial results |
| Track 3 |
Efficacy of adjuvant trastuzumab |
| Track 4 |
Adjuvant trastuzumab and cardiac safety |
| Track 5 |
Adjuvant dose-dense
AC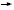 T with trastuzumab T with trastuzumab |
| Track 6 |
Coamplification of HER2 and topoisomerase II and benefit from anthracycline-based chemotherapy |
|
| Track 7 |
Utility of the Oncotype DX assay in patients with HER2-positive disease |
| Track 8 |
Use of adjuvant trastuzumab monotherapy |
| Track 9 |
Delayed adjuvant trastuzumab |
| Track 10 |
Incorporating adjuvant trastuzumab data into the Adjuvant! Online program |
| Track 11 |
Impact of tumor size and nodal status on the use of adjuvant trastuzumab |
|
|
Select Excerpts from the Interview
 Track 2
Track 2
 DR LOVE: George, would you provide an overview of the adjuvant
trastuzumab trials that were presented in 2005? DR LOVE: George, would you provide an overview of the adjuvant
trastuzumab trials that were presented in 2005? |
 DR SLEDGE: The two trials sponsored by the National Cancer Institute — NSABP-B-31 and NCCTG-N9831 — had somewhat overlapping schemas.
DR SLEDGE: The two trials sponsored by the National Cancer Institute — NSABP-B-31 and NCCTG-N9831 — had somewhat overlapping schemas.
In the control arm, patients received AC followed by paclitaxel. In the second arm, they began trastuzumab concurrent with paclitaxel and then continued trastuzumab for a total of one year. The N9831 trial also had a third arm in which patients began trastuzumab subsequent to the completion of taxane-based chemotherapy.
The BCIRG 006 trial, an international study, also had a control arm of AC followed by a taxane, but in this case it was docetaxel. In the second arm, trastuzumab was added to the taxane, and the study had an interesting third arm in which patients received carboplatin/docetaxel/trastuzumab but no anthracycline.
In the HERA trial, performed largely in Europe, patients had completed all of their systemic chemotherapy prior to being randomly assigned to an observation
arm versus one year of trastuzumab versus two years of trastuzumab.
 Track 3
Track 3
 DR LOVE: Can you summarize the efficacy data that have been reported? DR LOVE: Can you summarize the efficacy data that have been reported? |
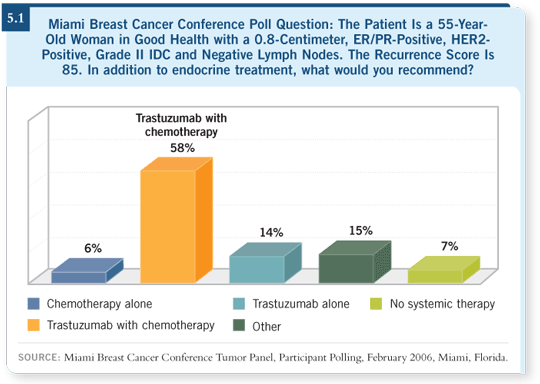
 DR SLEDGE: Data from the HERA trial comparing observation versus one year of trastuzumab show a significant benefit in the addition of trastuzumab, with a risk reduction of about 50% and a strikingly positive p-value (Piccart-Gebhart 2005). Interestingly, this trial included no specified chemotherapy regimen, and approximately one third of the patients had node-negative disease.
DR SLEDGE: Data from the HERA trial comparing observation versus one year of trastuzumab show a significant benefit in the addition of trastuzumab, with a risk reduction of about 50% and a strikingly positive p-value (Piccart-Gebhart 2005). Interestingly, this trial included no specified chemotherapy regimen, and approximately one third of the patients had node-negative disease.
In contrast to the HERA trial, early analysis of the N9831 trial demonstrated that the result from the sequential arm, in which trastuzumab was administered
after completion of chemotherapy, was not statistically significant, with a p-value of 0.01 (Romond 2005). From a purely statistical standpoint, this did not meet the boundaries required for early reporting.
The median follow-up in this trial is very short, and the number of events is small, so which regimen is better is still an unanswered question. In the arm in which trastuzumab was administered concurrently with chemotherapy, the result was highly significant.
In the BCIRG 006 trial, both of the trastuzumab-containing arms were superior to the nontrastuzumab-containing arm (Slamon 2005). The nonanthracycline
arm may be minimally inferior to the anthracycline-containing arm, but this is not yet a statistically significant difference and requires further follow-up.
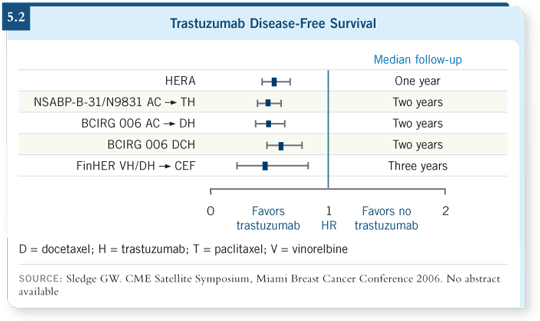
If we examine all these trials as a group and include the FinHER trial, a small Finnish trial of adjuvant trastuzumab (Joensuu 2005), in every single study we see significant benefits with the addition of trastuzumab to chemotherapy (5.2). As a result, trastuzumab has become the standard of care for patients receiving adjuvant therapy who have HER2-positive disease.
 Track 5
Track 5
 DR LOVE: We know from our Patterns of Care studies that the most
common chemotherapeutic regimen used in the United States for node-positive breast cancer is dose-dense AC followed by paclitaxel. That
regimen was not evaluated in any of the adjuvant trastuzumab trials from
which we have data, so physicians are questioning whether to use dose-dense AC DR LOVE: We know from our Patterns of Care studies that the most
common chemotherapeutic regimen used in the United States for node-positive breast cancer is dose-dense AC followed by paclitaxel. That
regimen was not evaluated in any of the adjuvant trastuzumab trials from
which we have data, so physicians are questioning whether to use dose-dense AC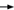 T with trastuzumab. Joyce, what are your thoughts? T with trastuzumab. Joyce, what are your thoughts? |
 DR O’SHAUGHNESSY: At the San Antonio Breast Cancer Symposium (2005),
the Memorial group reported on cardiac safety data from approximately 55
patients who received dose-dense AC followed by paclitaxel with trastuzumab
(Dang 2005).
DR O’SHAUGHNESSY: At the San Antonio Breast Cancer Symposium (2005),
the Memorial group reported on cardiac safety data from approximately 55
patients who received dose-dense AC followed by paclitaxel with trastuzumab
(Dang 2005).
Although some diminution occurred in LVEF, they saw no congestive heart
failure and no significant LVEF decline (5.3).
Cliff Hudis also presented data on the dose-dense AC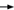 paclitaxel trial, INT-
C9741, reporting on five years of follow-up and showing no excess cardiac
toxicity when the anthracycline is given every two weeks, which is encouraging (Hudis 2005).
paclitaxel trial, INT-
C9741, reporting on five years of follow-up and showing no excess cardiac
toxicity when the anthracycline is given every two weeks, which is encouraging (Hudis 2005).
I am comfortable with dose-dense AC, and I administer the taxane the way it
was administered in the Intergroup or NSABP trials.
 DR LOVE: Peter, what do you think about the Memorial data?
DR LOVE: Peter, what do you think about the Memorial data?
 DR RAVDIN: The data from the Memorial Sloan-Kettering study are preliminary. The median follow-up was only about six months, so I would still say
that we don’t really know about the cardiac safety of that regimen.
DR RAVDIN: The data from the Memorial Sloan-Kettering study are preliminary. The median follow-up was only about six months, so I would still say
that we don’t really know about the cardiac safety of that regimen.
 Track 6
Track 6
 DR LOVE: George, Dennis Slamon presented a fascinating analysis of
topoisomerase II-alpha (TOPO II) amplification examined in the BCIRG
006 trial (Slamon 2005; Press 2005). Can you comment on the data and
whether you think they are clinically applicable at this time? DR LOVE: George, Dennis Slamon presented a fascinating analysis of
topoisomerase II-alpha (TOPO II) amplification examined in the BCIRG
006 trial (Slamon 2005; Press 2005). Can you comment on the data and
whether you think they are clinically applicable at this time? |
 DR SLEDGE: TOPO II is one of the molecular targets for doxorubicin. It’s
found on chromosome 17, right next to where HER2 is located, so it’s not
uncommon for them to be coamplified.
DR SLEDGE: TOPO II is one of the molecular targets for doxorubicin. It’s
found on chromosome 17, right next to where HER2 is located, so it’s not
uncommon for them to be coamplified.
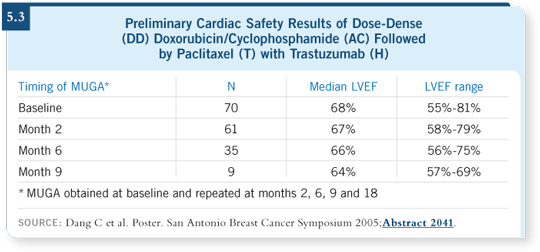
We know from the BCIRG trial, as a result of careful analysis for TOPO II
and HER2, that approximately a third of the breast cancers that are HER2
amplified by FISH are also amplified for TOPO II by FISH. A preliminary
analysis of the data suggests that patients who benefit from an anthracycline/trastuzumab-containing regimen tend to be the patients who have the
coamplification of TOPO II and HER2 (5.4).
Clearly, these data are preliminary, but they’re fascinating and make sense
biologically. Hopefully, in another year or two we’ll have sufficient follow-up
data to point us in one direction or another.
 Track 11
Track 11
 DR LOVE: George, for a patient with a one- to two-centimeter,
ER-positive, HER2-positive tumor, are you likely to recommend
trastuzumab? DR LOVE: George, for a patient with a one- to two-centimeter,
ER-positive, HER2-positive tumor, are you likely to recommend
trastuzumab? |
 DR SLEDGE: Yes. That patient falls within the eligibility criteria of the
HERA trial, so we have clinical trial data that say it’s appropriate to use
trastuzumab (Piccart-Gebhart 2005).
DR SLEDGE: Yes. That patient falls within the eligibility criteria of the
HERA trial, so we have clinical trial data that say it’s appropriate to use
trastuzumab (Piccart-Gebhart 2005).
 DR LOVE: Do you use trastuzumab if the tumor is less than one centimeter in
size?
DR LOVE: Do you use trastuzumab if the tumor is less than one centimeter in
size?
 DR SLEDGE: Right or wrong, I don’t. These trials included very few patients
with tumors in the subcentimeter category.
DR SLEDGE: Right or wrong, I don’t. These trials included very few patients
with tumors in the subcentimeter category.
 DR LOVE: Joyce, how does tumor size affect your decision to use adjuvant
trastuzumab for a patient with HER2-positive, node-negative breast cancer?
DR LOVE: Joyce, how does tumor size affect your decision to use adjuvant
trastuzumab for a patient with HER2-positive, node-negative breast cancer?
 DR O’SHAUGHNESSY: If the tumor is greater than a centimeter in size and the
patient has no contraindications to trastuzumab, I recommend it. For patients
with tumors less than a centimeter, it depends on what I think their residual
risk will be. For example, if a tumor is eight or nine millimeters but ER/PR-negative, Grade III and HER2-positive by FISH, I recommend trastuzumab.
DR O’SHAUGHNESSY: If the tumor is greater than a centimeter in size and the
patient has no contraindications to trastuzumab, I recommend it. For patients
with tumors less than a centimeter, it depends on what I think their residual
risk will be. For example, if a tumor is eight or nine millimeters but ER/PR-negative, Grade III and HER2-positive by FISH, I recommend trastuzumab.
For patients at higher risk, I always use AC followed by paclitaxel or docetaxel
with trastuzumab, but for the patients at lower risk, I consider TCH because it
has less cardiac toxicity.
Select publications

