
| Tracks 1-10 |
| Track 1 |
Introduction |
| Track 2 |
Impact of margin status on
re-excision and local control |
| Track 3 |
Partial breast irradiation techniques evaluated in clinical trials |
| Track 4 |
Postlumpectomy radiation therapy in women over age 70 |
| Track 5 |
Postlumpectomy radiation therapy and hormonal therapy for women with DCIS |
|
| Track 6 |
Perspective on the role of aromatase inhibitors in the prevention and adjuvant settings |
| Track 7 |
Utility of the Oncotype DX assay in clinical practice |
| Track 8 |
Clinical indications for the use of sentinel lymph node biopsy |
| Track 9 |
Axillary dissection after identification
of a positive sentinel node |
| Track 10 |
False-negative rates in sentinel lymph node biopsies |
|
|
Select Excerpts from the Interview
 Track 5
Track 5
 DR LOVE: How are you treating patients with DCIS in terms of endocrine therapy? DR LOVE: How are you treating patients with DCIS in terms of endocrine therapy? |
 DR BORGEN: We have viewed tamoxifen as a highly appropriate option for treating a patient with ER-positive DCIS since the NSABP-B-24 trial (Fisher 1999).
DR BORGEN: We have viewed tamoxifen as a highly appropriate option for treating a patient with ER-positive DCIS since the NSABP-B-24 trial (Fisher 1999).
However, when we consider risks, benefits and quality-of-life issues, it’s common for our New York patients to demur, so we probably have one of the lowest percentages of patients with ER-positive DCIS on tamoxifen in the country. The same can be seen in our prevention setting, in which we’ve not been successful in getting patients to take tamoxifen.
 DR LOVE: What are the concerns about tamoxifen in these settings?
DR LOVE: What are the concerns about tamoxifen in these settings?
 DR BORGEN: The two most obvious concerns are endometrial cancer and gynecological events. Even when we provide the raw numbers on how infrequent
those events are, because we are talking about minimal, if any, impact on long-term survivorship and moderate impact on local control, it simply is not an attractive option.
DR BORGEN: The two most obvious concerns are endometrial cancer and gynecological events. Even when we provide the raw numbers on how infrequent
those events are, because we are talking about minimal, if any, impact on long-term survivorship and moderate impact on local control, it simply is not an attractive option.
 DR LOVE: For a postmenopausal patient with DCIS who is interested in endocrine therapy but finds tamoxifen intolerable because of side effects, do you offer an aromatase inhibitor?
DR LOVE: For a postmenopausal patient with DCIS who is interested in endocrine therapy but finds tamoxifen intolerable because of side effects, do you offer an aromatase inhibitor?
 DR BORGEN: We’d like more information about DCIS and aromatase inhibitors,
but since the initial publication of the ATAC data (Baum 2002), aromatase
inhibitors have become our endocrine therapy of choice for postmenopausal
patients with ER-positive, invasive cancers.
DR BORGEN: We’d like more information about DCIS and aromatase inhibitors,
but since the initial publication of the ATAC data (Baum 2002), aromatase
inhibitors have become our endocrine therapy of choice for postmenopausal
patients with ER-positive, invasive cancers.
That literally happened overnight, like gangbusters, and so a “bleed over” to postmenopausal patients with DCIS is natural.
 Track 6
Track 6
 DR LOVE: If clinical research data demonstrate a superior antitumor effect and a better toxicity profile with aromatase inhibitors versus tamoxifen, how do you think these agents will be accepted in the prevention and adjuvant settings? DR LOVE: If clinical research data demonstrate a superior antitumor effect and a better toxicity profile with aromatase inhibitors versus tamoxifen, how do you think these agents will be accepted in the prevention and adjuvant settings? |
 DR BORGEN: In my clinical practice, it’s clear that the aromatase inhibitors are vastly better tolerated than tamoxifen in postmenopausal patients. Our surgeons are beginning to give first-line endocrine therapy without a mandatory
consult from medical oncology. This was a policy change at Memorial a few years ago. Our oncologists were overwhelmed by the volume of invasive carcinomas, so the surgeons took a front-line role. We perform bone density tests before we start our patients on aromatase inhibitors, and treating these patients has been satisfying.
DR BORGEN: In my clinical practice, it’s clear that the aromatase inhibitors are vastly better tolerated than tamoxifen in postmenopausal patients. Our surgeons are beginning to give first-line endocrine therapy without a mandatory
consult from medical oncology. This was a policy change at Memorial a few years ago. Our oncologists were overwhelmed by the volume of invasive carcinomas, so the surgeons took a front-line role. We perform bone density tests before we start our patients on aromatase inhibitors, and treating these patients has been satisfying.
 DR LOVE: Surgeons, particularly breast cancer surgeons, used to prescribe tamoxifen routinely. It is interesting that, when the data began to favor the aromatase inhibitors, I saw surgeons hesitate in terms of treating patients with these agents. Do you find that is changing and surgeons are now using aromatase
inhibitors?
DR LOVE: Surgeons, particularly breast cancer surgeons, used to prescribe tamoxifen routinely. It is interesting that, when the data began to favor the aromatase inhibitors, I saw surgeons hesitate in terms of treating patients with these agents. Do you find that is changing and surgeons are now using aromatase
inhibitors?
 DR BORGEN: Tamoxifen has a 40-year head start, and surgeons are sometimes slow to change, but it’s definitely changing. We’ve treated a growing number of older patients with larger, ER-positive tumors with neoadjuvant aromatase inhibitors and seen some striking results.
DR BORGEN: Tamoxifen has a 40-year head start, and surgeons are sometimes slow to change, but it’s definitely changing. We’ve treated a growing number of older patients with larger, ER-positive tumors with neoadjuvant aromatase inhibitors and seen some striking results.
We can downsize and downstage tumors for the majority of these patients, which leads to a far smaller surgical procedure, and in some cases the
tumors go away completely, and we are left with following a patient
closely. I believe the aromatase inhibitors belong in a breast surgeon’s practice and armamentarium.
 DR LOVE: Have you used neoadjuvant aromatase inhibitors in your practice or in a clinical trial setting?
DR LOVE: Have you used neoadjuvant aromatase inhibitors in your practice or in a clinical trial setting?
 DR BORGEN: Both. We were impressed by the initial work that Mike Dixon
published from the United Kingdom (Dixon 1999, 2000, 2001; [4.1]). We
conducted a small in-house study and corroborated his findings exactly, both
in terms of tumor response rates and patient acceptance.
DR BORGEN: Both. We were impressed by the initial work that Mike Dixon
published from the United Kingdom (Dixon 1999, 2000, 2001; [4.1]). We
conducted a small in-house study and corroborated his findings exactly, both
in terms of tumor response rates and patient acceptance.
 DR LOVE: Have you used neoadjuvant aromatase inhibitors to downsize
tumors to convert a mastectomy to a lumpectomy?
DR LOVE: Have you used neoadjuvant aromatase inhibitors to downsize
tumors to convert a mastectomy to a lumpectomy?
 DR BORGEN: Absolutely. Certainly in the older patient population we have
done that. I would qualify that by saying it’s not so much to convert a mastectomy to a lumpectomy as it is to downstage the disease.
DR BORGEN: Absolutely. Certainly in the older patient population we have
done that. I would qualify that by saying it’s not so much to convert a mastectomy to a lumpectomy as it is to downstage the disease.

 Track 9
Track 9
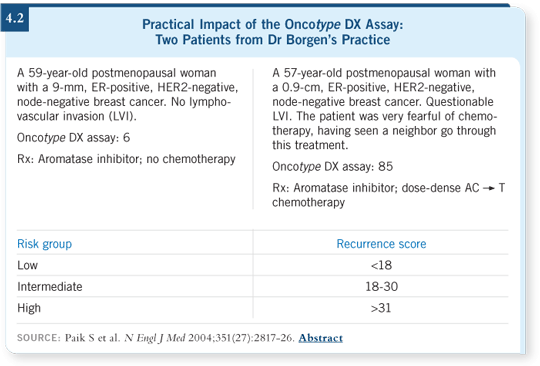
 DR LOVE: What is your opinion of the Oncotype DX assay (4.2), and how
do you see it being used clinically? DR LOVE: What is your opinion of the Oncotype DX assay (4.2), and how
do you see it being used clinically? |
 DR BORGEN: We’re excited about the possibility of a truly genomic approach
to the disease. We use the Oncotype DX assay in borderline cases in which a
low recurrence score would preclude cytotoxic chemotherapy. For the patient
who has a larger tumor, a higher-grade tumor or other mitigating factors,
we’re not using Oncotype DX as a sole factor in precluding chemotherapy, but
in those borderline cases it’s been enormously helpful.
DR BORGEN: We’re excited about the possibility of a truly genomic approach
to the disease. We use the Oncotype DX assay in borderline cases in which a
low recurrence score would preclude cytotoxic chemotherapy. For the patient
who has a larger tumor, a higher-grade tumor or other mitigating factors,
we’re not using Oncotype DX as a sole factor in precluding chemotherapy, but
in those borderline cases it’s been enormously helpful.
 DR LOVE: Medicare is now paying for the Oncotype DX assay. Do you think
that is tied to the fact that it appears to be cost effective?
DR LOVE: Medicare is now paying for the Oncotype DX assay. Do you think
that is tied to the fact that it appears to be cost effective?
 DR BORGEN: That’s correct. If we eliminated chemotherapy for one third
of small, node-negative breast cancers — and the estimates could be higher
than one third — then this would look like the best money ever spent on a
medical test.
DR BORGEN: That’s correct. If we eliminated chemotherapy for one third
of small, node-negative breast cancers — and the estimates could be higher
than one third — then this would look like the best money ever spent on a
medical test.
Select publications

